> Запросить цену! <
Модель шприцевого насоса Injectomat MC Agilia имеет расширенный набор функций (программирование скорости введения дозы, введение нагрузочной дозы, программирование болюса, графическая история и др.), предназначен для использования в отделениях интенсивной терапии и других специализированных отделениях.
- Описание
- Производитель
- Отзывы (0)
- Доставка
- Вопрос-ответ
Описание
Варианты программирования инфузии:
- программирование скорости в мл/ч
- программирование в единицах дозы (например, мкг/кг/мин) – насос автоматически пересчитает скорость в мл/ч
- программирование введения определенного объема или дозы за определенное время
- программирование объема и скорости инфузии
- Возможность программирования введения нагрузочной дозы/дозы индукции
Характеристики насоса
| Диапазон скорости введения | от 0,1 до 1200 мл/ч (зависит от объема шприца) |
|---|---|
| Диапазон скорости введения болюса | от 50 до 1200 мл/ч. Возможно как ручное, так и программируемое введение болюса |
| Варианты объемов шприцев | 5, 10, 20, 30/35, 50/60 мл |
| Программируемая пауза | от 1 мин до 24 ч |
| Журнал | 1500 записей |
| Встроенная батарея | NiMH, время работы от батареи 12 часов при скорости инфузии 5 мл/ч |
| Размеры | 345 мм х 135 мм х 170 мм / 2,15 кг |
Другие преимущества и особенности
- В память загружены характеристики 50 моделей шприцев разных производителей, что позволяет учесть их особенности и обеспечить высокую точность при проведении инфузии
- При установке шприца в насос перемещение толкателя и фиксация поршня осуществляется вручную – нет задержек времени
- Защита поршня – защитная рамка предотвращает непреднамеренное введение болюса при случайном ударе по толкателю поршня, например при падении насоса или при транспортировке
- Большой информативный экран – достаточно беглого взгляда, чтобы быстро оценить состояние инфузии
- Отражение на экране названия вводимого раствора/препарата
- Возможность установки библиотеки лекарственных препаратов Vigilant® Drug Lib. В ней для каждого препарата программируется ряд параметров: название, мягкие пределы (наиболее высокая используемая скорость введения), жесткие пределы (максимально допустимая скорость введения), допустимые режимы инфузии, что значительно повышает безопасность при проведении инфузии
- Режим поддержания вены открытой (ПВО) – после завершения инфузии насос автоматически продолжит вводить раствор с небольшой скоростью (от 1 до 5 мл/ч) с целью поддержания проходимости венозного доступа
- Динамическая система контроля давления – реагирует на стабильное изменение уровня давления в системе (до достижения заданного предела), позволяет быстро обнаружить окклюзию или отсоединение системы и своевременно принять необходимые меры
- Графическое и цифровое отражение значения давления в системе
- Графическое отражение истории: объем/введенная доза, давление, скорость
- Поворачиваемый зажим позволяет крепить насос в необходимом положении
- Возможность объединения насосов друг с другом
- Ночной режим – сниженная яркость, выключение звука кнопок; автоматический переход в ночной режим в заданное время
- Сервисное обслуживание – один раз в три года
Комплектация
| Насос Injectomat MC Agilia | 1 шт |
| Адаптер питания | 1 шт |
| Сетевой адаптер | 1 шт |
| Гарантийный талон | 1 шт |
Информация о поставке
ООО “МЕДПРИБОР ПРО” – официальный поставщик оборудования Fresenius Kabi в России.
Для получения коммерческого предложения, документации и авторизации поставки пришлите свой запрос на почту info@medpribor.pro
Производитель
| Производитель / Бренд |
Fresenius Kabi |
|---|---|
| Страна производства |
Германия |
| Гарантия |
Да |
| Модель |
Z018156 |
Отзывы (0)
Доставка
СПОСОБЫ ДОСТАВКИ:
- экспресс доставка в течении 5 часов собственным транспортом компании (Москва и область)
- курьерской службой или ТК по РФ и торговому союзу (до двери)
- до пунктов самовывоза
СПОСОБЫ ОПЛАТЫ: при получении или предоплата по счету
БЕСПЛАТНАЯ И ПЛАТНАЯ ДОСТАВКА:
- Бесплатная доставка медицинского оборудования при заказе от 10.000 рублей осуществляется по Москве и МО
- Бесплатная доставка медицинского оборудования при заказе от 60.000 рублей осуществляется по всей России
- Платная доставка заказов до 60.000 руб. осуществляется по всей России *
- Отправка заказов любой стоимостью в страны торгового союза осуществляется платно (в Казахстан, Беларусь и другие страны).
* Стоимость доставки до 60000 руб. рассчитывается в соответсвии с тарифами служб доставки (см. раздел доставки). Стоимость доставки зависит от габаритов товара и веса, подробный расчет вы можете получить у менеджеров интернет-магазина.
ДЛЯ КОНТРАКТНЫХ КЛИЕНТОВ: Мы осуществляем поставки медицинского оборудования оптом, в розницу и на тендерной основе. Для больниц, поликлиник, фондов и других организаций доставка, монтаж и обучение может быть включено в стоимость товара. (по желанию заказчика)
Вопрос-ответ
Вопросы и ответы покупателей
Еще не задано вопросов, будьте первым, кто спросит о товаре.
Похожие товары
Шприцевой насос Injectomat® MC Agilia — Fresenius Kabi
- Описание
Описание
Шприцевой насос Injectomat MC Agilia обладает расширенным набором функций и является оптимальным выбором для использования в отделениях реанимации и интенсивной терапии.
- Эргономичный дизайн и дружественный интерфейс
- Большой информативный экран, хорошо читаемый с расстояния 3–5 м
- Варианты программирования инфузии:
- программирование в единицах дозы (мкг/кг/мин, Ед/ч и т.д.) – перерасчет в мл/ч осуществляется автоматически
- программирование скорости инфузии в мл/ч
- программирование введения заданного объема за заданное время
- программирование введения заданного объема с заданной скоростью
- Широкий выбор единиц для программирования скорости инфузии: нг/мкг/мг/Ед/кЕд/мкал/ кал/ккал/мкмоль/ммоль/моль на кг в мин/ч/сут
- Быстрая подготовка к инфузии – установка шприца в насос полностью осуществляется вручную
- Наличие режима микроинфузии, в котором скорость программируется с шагом 0,01 мл/ч
- Программируемое введение болюса (точное введение объема/дозы с заданной скоростью)
- Широкий набор функций, обеспечивающих безопасность инфузии и снижение нагрузки на персонал
- Защита поршня и толкателя — защитная рамка предотвращает непреднамеренное введение болюса и/или повреждение толкателя при случайном ударе по нему
- Возможность установки библиотеки лекарственных препаратов Vigilant® Drug Lib, в которой для лекарственного препарата задаются типичные параметры инфузии, допустимые режимы, пределы скорости инфузии и введения болюса, что значительно ускоряет подготовку к инфузии и обеспечивает высочайшую безопасность, исключая риск случайной передозировки
- Динамическая система контроля давления позволяет быстро обнаружить окклюзию или отсоединение системы и своевременно принять необходимые меры
- Встроенная ручка для удобной переноски и объединения насосов друг с другом
- Ночной режим – сниженная яркость экрана, выключение звука кнопок; возможность настройки автоматического перехода в ночной режим и выхода из него в заданное время
-
Syringe PumpInstructions for Use
Injectomat Agilia
-
21. INTRODUCTION
…………………………………………………………………………………………………………………………
4Intended use
……………………………………………………………………………………………………………………………….
4Precautions to be taken
………………………………………………………………………………………………………………..
42. DESCRIPTION OF INJECTOMAT AGILIA
…………………………………………………………………………………..
53. OPERATIONS FOR
USE……………………………………………………………………………………………………………..
74. DISPLAY AND
SYMBOLS………………………………………………………………………………………………………….
125. ALARM AND SAF
6. MENU ………………Permanent menu .Menu to be selecte
7. OPTION……………
8. DRUGS…………….Introduction ……….Infusion mode:
VolDisplay and symboSafety features andService Options …Drug
library ……….9. USER TEST………
10. PERFORMANCESRates range ………Volume Limit
……..Volume/Time……..Syringe list ………..Accuracy
…………..Programmable pauPressure managemOcclusion alarm
re11. TECHNICAL CHAElectrical powers..Battery
……………..Communication poInfrared
communicCompliance……….Dimensions — WeigTrumpet curves….12. GUIDANCE AND MElectromagnetic emElectromagnetic
imElectromagnetic imRecommended seInjectomat AgiliaContents2309-11_nu_injectomat_agilia_INT
ETY FEATURES
…………………………………………………………………………………………….
14………………………………………………………………………………………………………………………
16………………………………………………………………………………………………………………………
16d in option
mode……………………………………………………………………………………………..
17………………………………………………………………………………………………………………………
18………………………………………………………………………………………………………………………
20………………………………………………………………………………………………………………………
20ume/time and limit volume
………………………………………………………………………………..
21ls…………………………………………………………………………………………………………………..
23 warnings
………………………………………………………………………………………………………
23………………………………………………………………………………………………………………………
23………………………………………………………………………………………………………………………
24………………………………………………………………………………………………………………………
25…………………………………………………………………………………………………………………….
26………………………………………………………………………………………………………………………
26………………………………………………………………………………………………………………………
26………………………………………………………………………………………………………………………
26………………………………………………………………………………………………………………………
27………………………………………………………………………………………………………………………
27se
…………………………………………………………………………………………………………………
27ent
……………………………………………………………………………………………………………….
28sponse time and Bolus volume at occlusion release
……………………………………………. 29RACTERISTICS………………………………………………………………………………………………
30………………………………………………………………………………………………………………………
30………………………………………………………………………………………………………………………
30rt……………………………………………………………………………………………………………………
30ation
………………………………………………………………………………………………………………
30………………………………………………………………………………………………………………………
30ht…………………………………………………………………………………………………………………..
31………………………………………………………………………………………………………………………
31ANUFACTURERS DECLARATION ON EMC
………………………………………………….
32issions — Table
201…………………………………………………………………………………………
32munity — Table 202
………………………………………………………………………………………….
32munity — Table 204
………………………………………………………………………………………….
33paration distances between portable and mobile RF communication
equipment and — Table
206…………………………………………………………………………………………………….
34 -
2309-11_nu_injectomat_agilia
13. CLEANING AND USE CONDITIONS
…………………………………………………………………………………………..
35Cleaning and
disinfecting…………………………………………………………………………………………………………….
35Environmental
conditions…………………………………………………………………………………………………………….
35Use of the internal
battery……………………………………………………………………………………………………………
35Recommendations……………………………………………………………………………………………………………………..
3614.
SERVICES………………………………………………………………………………………………………………………………..
37Conditions of guarQuality control……Preventive
maintenServicing …………..Data racks, access_INT 3antee………………………………………………………………………………………………………………
37………………………………………………………………………………………………………………………
37ance
……………………………………………………………………………………………………………..
37………………………………………………………………………………………………………………………
37ories and maintenance tools
…………………………………………………………………………….
38 -
41. Introduction
Injectomat Agilia is the first syringe pump of the Agilia range,
our new generation of infusion pump.It is simple to use and
training is fast thanks to its ergonomics and interactive
screens.Very intuitive, Injectomat Agilia was also developed to
promote safety. The pusher protection «Push-guard»offers a maximum
safety in case the pump falls.The monitoring of pressure as well as
many safety features optimize the operation of Injectomat
Agilia.Robust and adaptable thanks to a wide choice of options,
Injectomat Agilia makes it possible to answer thewhole of your
needs.Intended use Injectomat Agilia is This pump must be
Precautions to The symbol vis Injectomat Agilia wof the medical
devicesradiations avoids the uis placed near devicedistances
between equ The device must noshould always be used The device can
be etc. If you wish to use tmust be used in a horiz The
physiological esyringe. Check that thealarm setting times in r In
case of unexpecalarm, to stop infusionChapter 6). In case whthe
user should consid2309-11_nu_injectomat_agilia_INTa syringe pump for intravenous drug infusion. used by
professionals working in hospitals.be takenible on the device, recommends this user guide should be
completely read.as tested in accordance with the applicable
standards of electromagnetic compatibility. Its immunity makes it possible to ensure correct operation.
The limitation of the emittedndesirable interference with other
equipments such EEG, ECG, If Injectomat Agilias like surgical
equipment HF, X-rays, NMR, mobile phones, Wifi points…,
minimalipment are essential (see page 33).t be used in presence of
inflammable anaesthetic agents due to a risk of explosion. It away
from all risk areas.disturbed by pressure or pressure variations,
mechanical shocks, heat ignition sources,he devices in a specific
condition, please contact our After-Sales Department. The pumpontal
and stable position to work correctly.ffects of medicine can be influenced by the characteristics of
the device and disposabley are compatible with prescriptions, the
characteristics of trumpet curves and occlusionelation to the
programmed flow rate.ted situation in the pump controls or
environment, the state of the art safe-design is to and to display
an error code. The user is invited to be aware of those alarms
(seeere the device is used to deliver life sustaining therapies,
like short half-life medications,er adequate provisions for back-up
therapy delivery solutions. -
2309-11_nu_injectomat_agilia
2. Description of Injectomat Agilia
1 — Syringe barrel cl2 — Syringe flange c3 — Pusher4 —
«Push-guard»10_INT 5
asp 5 — Handle 9 — Mains connectionradle 6 — Assembly bolt 10 —
Fixing button7 — Infrared cell 11 — «Swinglock clamp»8 — Communication
portand DC power input-output
11 9 8
7
65
1 2 3 4
-
612 — Mains warning13 — Screen14 — Silence Alarm15 — ON/OFF16 —
Bolus or Prime15 16
122309-11_nu_injectomat_agilia_INT
17 — 22 — Validationto Value selection 23 — Stop: infusion
stop20 — 24 — Menu / Exit21 — Functioning, pre-alarm
and alarm warnings
17 1918
24232221
20
13 14
-
2309-11_nu_injectomat_agilia
3. Operations for use
Installation of Injectomat Agilia
Injectomat Agilia can be used on a table, pole or rail.
On table
Two » Injectomat Agassembled together _INT 7
On pole On rail
ilia » maximum can be during infusion
Three devices maximum can be assembled on a pole or during
transportationWhen the devices are assembled, the assembly bolts must be in
closed position.3 pumps on a pole: at least 2 fixing clamps must be locked.
-
8Using the fixing clamp
The swinglock clamp is only orientable when closed against the
pump. It is maintained in its vertical or horizontal position with
the fixing button. The following images show how to modify the pump
installation, from a pole to a rail position.Unscrew the clamdevice from the p
Rotate the fixing c90 degrees.
A
B2309-11_nu_injectomat_agilia_INT
p screw (A) and disengage the ole. Push the fixing button
(B).Fold the fixing clamp against the pump. This is the recommended
position for the swinglock clamp when the device is placed on a
flat surface.lamp downward through Move the fixing clamp outward (A). The
fixing button is released automatically. Engage the device on the
rail and use the clamp screw (B) to secure it.BA
-
2309-11_nu_injectomat_agilia
Installing a syringe (patient not connected)
Connect the extension set to the syringe according to proper
practices.Check that there is no air bubble left in the
syringe.Place the syringe in its cradle, the flanges correctly inserted
in the provided slot . Secure the syringe with the syringe barrel
clasp.Move the pusher f_INT 9
orward to the syringe head. Check the general installation.
-
10
1 — Operations for use 2 — Syringe selection
Check Injectomat Connect the powersource and to the syrinlights
up. Press the ke For the first startupinternal battery. Press the
kpump OFF. To disconsupply, disconnect maunplugging the device3 — Prime
Connect the extens Check the patient i To start the prime, pone
short press, then air bubbles are elimina To stop the prime,
Connect the patienNote: During priming, set to its maximum
va2309-11_nu_injectomat_agilia_INTAgilia is not damaged. supply cord to the main ge pump: the
mains warningy to turn the pump ON., refer on page 35: Use of the
ey continuously to turn the nect the device from mains ins-wise
plug first before power inlet.The installed syringe must correspond to the syringe displayed.
OK: to confirm syringe,or, C: to change syringe selection then
confirm.4 — Flow rate selection/start
ion set to the syringe.s not connected.ress the key twice:
one continuous press until all ted from the line.
release the key.t.the occlusion pressure level is lue (900
mmHg).Select flow rate. Check the infusion parameters (syringe, flow
rate, ). Start: press the key to start infusion. -
2309-11_nu_injectomat_agilia
Silence alarm Access to the menu
Press the key until the flow rate
te (ml/h) and confirm.e occlusion pressure level is set (900
mmHg).Press the key: the infusion stops. Start infusion pressing the
key. Pause programming: press the key twice. Select the pause
duration. The pause can also be programmed from the menu. When the
pause duration is finished, press the key to start infusion.tamnguyenHighlight
tamnguyenHighlight
-
12
4. Display and symbols
Injectomat Agilia displays the infusion parameters in progress
through specific symbols.Continuous display
Infusion in progressMain indicator lights provide information on
status of the infusion in progress.Pause The symbol flashes when activated.
Indicator lights
Help
Alarms and safeties features
or2309-11_nu_injectomat_agilia_INT
Battery lifeAppears when the device is operating on
battery.Three different levels of charge are symbolized.Mainsconstant yellow
Main indicator lights provide information on status of the
infusion in progress.Infusion in progressflashing green
Pre-alarmflashing orange
Alarmflashing red
Start
These symbols help the user in programming the pump.
Validation
Access to function
Previous screen
Cancel
Change syringe selection
Selected
Not selected
Mains disconnection alarm
Main symbols for alarm and safety features.Pressure increase
Pressure drop
-
2309-11_nu_injectomat_agilia
Selection keys
Fast increment key
Increment key
MENU_INT 13
Keys for selection of flow rate(ml/h), volume limit (ml),
values.Decrement key
Fast decrement key
Fast access to maximum values +
Fast access to minimum values +
Infused volume
The menu is dedicated to infusion options that are selected by
the user.Battery life
Keyboard locking
Maintenance
Date/Hour
Data event log
Syringe
Sound level
Volume limit
Volume/Time
Pause
Pressure
Night mode
-
14
5. Alarm and safety features
Injectomat Agilia has a continuous inspection system that
functions as soon as the pump is in use. Visualmessages are
displayed to understand the alarm cause. Press on to silence alarm
according to tablebelow.Control Visual message Infusion stop
Silence alarm Activation
Battery
BATTERPRE-ALA
BATTER
Mains POWER DISCON
Installed syringe
SYRINGINSTALL
Infusion
END OF PRE-ALA
END OF ALARM
Volume Limit
END OF VOLUMEPREALA
END OF VOLUMEThe max2309-11_nu_injectomat_agilia_INT
Y RM
NO YES Low battery. Note: Battery alarm activated when at least
30 minutes battery life remaining. (If the battery has previously
been charged).Y ALARM YES YES (2 min) Discharged battery.Note: The pump will
turn OFF automatically within 5 minutes. Connect the pump to the
mains.NECTIONNO YES Mains disconnection. (Alarm Selection :
refer [Par 13], page 19.E ATION
YES YES (2 min) Pusher or syringe barrel clasp or flange
detection.YES YES (2 min) Syringe not correctly installed.Note: The alarm
goes OFF as soon as the installation is correct. A silence alarm of
2 min is automatically activated when the pump is switched on.INFUSION RM
NO YES The pre-alarm is triggered when the time before end of
infusion is less than 5 min and the remaining volume in the syringe
is less than 10% of the syringe capacity.INFUSION YES YES Empty syringe.
LIMIT RM
NO YES The pre-alarm is triggered when the time before end of
volume limit is less than 5 min and the remaining volume in the
syringe is less than 10% of the syringe capacity.LIMIT ALARM
Stop/KVO/continuous
YES (*) Limit volume reached.(*) Silence duration for KVO: page
18imum volume that may be infused under single fault condition is
1 ml.tamnguyenHighlight
-
2309-11_nu_injectomat_agilia
V/T
END OF VOL./TIME PREALARM
NO YES 5 minutes before V/T alarm or 10% of the total syringe
capacity.END OF VOL./TIME ALARM
Stop/KVO/ YES (*) V/T limit reached.
Pressure
OCCLUSPRE-ALAOCCLUS
PRESSU
PRESSUINCREA
Other alarms
PLUNGEALARMDISENGMECHANALARMFLASHINRATE
NO VALI
Audible
STOP m
Er — mes(Er01, ErIn case ostop the persists wtechnicia
Control Visual message Infusion stop
Silence alarm Activation_INT 15
continuous mode
(*) Silence duration for KVO: page 18
ION RM
NO YES — 50 mmHg from the programmed limit.
ION ALARM YES YES (2 min) Programmed limit reached.
RE DROP NO YES Pressure drop in the infusion line.(This alarm
can be selected in options).RE SE
NO YES Pressure increase in the infusion line.(This alarm can be
selected in options).R HEAD YES YES Pusher incorrectly inserted.
AGEMENT ISM
YES YES Disengaged mechanism.
G FLOW NO — Flashing starts 3 seconds after no confirmation of
selection. An audible alarm is activated 15 seconds afterwards.DATION NO YES Flashing starts 3 seconds after no confirmation of
selection. An audible alarm is activated 15 seconds afterwards.signal — — No syringe selection > 2 minutes.
— — Unauthorized key.
essage — — End of pause duration.
sage 02)
YES — Technical alarm. Press the key.
f malfunction alarm, note the error message (ErXX). Disconnect
from the mains and device by pressing the OFF key (10 — 15 seconds
can be necessary). If the alarmhen the device is switched on again, without use on patient,
contact the qualified ns in your establishment or our After-Sales
Department. -
16
6. Menu
Permanent me
Operation Key
Access menu/previous menu:
Select with:
Confirm with:
Selected / Not sel
FunctionInfused volume
Pressure
Battery life
Pause
Locking
SyringeDisplay only if [Par 15] «syringe display» not
selected2309-11_nu_injectomat_agilia_INTnu
___
ected
Description Operation SymbolDisplay of infused volume and total
infused volume resetClear the infused volume
Pressure limit adjustment and DPS mode activation
Pressure limit DPS mode activation
Battery life display Display in hour and minute for a selected
ratePause duration adjustment
Hours and minutes adjustment
Keyboard locking and unlocking
LockingThe and keys are never locked.
Brand capacity and type of the used syringe
Syringe used
-
2309-11_nu_injectomat_agilia
Menu to be selected in option modeFunction Description Operation
SymbolVolume/TimeFunction accessible in STOP mode only
Volume/time programming Volume Time End VTI (stop, KVO,
continuous)Maintenance Information on Maintenance date
Data Event logFunction accessible in STOP mode onlySound
levelVolume limitFunction accessible in STOP mode
Date/hour
Night modeOnly if «Manual mode» selected in Ward option [Par
18]CAUTION: the menu_INT 17
maintenance, version, functioning duration, etc.
SN (serial number) Software version
Up to 1500 events recorded
Syringe Pressure limit Flow rate
Audible level adjustment 7 accessible levels
Volume limit programming Select VL or OFF End VL (stop, KVO,
continuous)Date and hour dd/mm/yyyy h/minManual Mode change:night/day or
day/nightManual mode interrupts auto-mode. Night mode is re-activated on
next defined night cycle [Par 18]can change depending on the selected infusion options.
-
18
7. Option
Operation Key
Options access: when device is turned off
Option selection:
Confirm by:
Previous menu
Selected / Not sel
Selected values on us
Option FuUser [Util 1] Sc
Display ofsymbols
[Util 2] MeInsertion ooptions in
[Util 3] Co[Util 4] PrPressure mDynamic PSystem
[Util 5] KV(Keep Vei
[Util 7] Da[Util 8] La2309-11_nu_injectomat_agilia_INT
___
ected
e are memorized when the device is turned OFF at end of
programming.nction Choice Description / reen options the different
Battery Permanent display of battery symbol Pressure Display of
pressure symbol Volume info. (or) Display of infused volume Time
info. (or) Display of remaining time of infusion Battery life info.
(or) Display of battery life Man Choice of symbol «infusion inprogress» Moonnu options f the different the menu
Volume limit Volume limit selection Volume/time V/T selection
Sound level Audible signal selection Maintenance Maintenance
selection Data event log Display of event log Date/hour Date/hour
selectionntrast Screen contrast adjustmentessure
ode Variable mode (with maximum and limit)
3 level mode (with thresholds and limit)
ressure DPS with drop threshold and pressure increase
thresholdNo DPS mode activation
On Open)
KVO1: OFF, 0.1 to 5 ml/h KVO2: OFF, 0.1 to 5 ml/h Continuous:
YES/NOSilence duration: delay for end of V/T or end of VL
re-activation alarm (60 minutes maximum)Continuous mode: at the end of V/T or VL mode, infusion
continues at the current selected rate.te/hour Date selection: dd/mm/yyyy Hour selection: h/minnguage
Franais / EnglishtamnguyenHighlight
tamnguyenHighlight
-
2309-11_nu_injectomat_agilia
Option Functions Option choiceWard Ward code Code: 0000 (default
code: 0200).[Par 1] Beep level 1 tonality 2 tonalities Key Bip Preventive
silence : refer page 11 «Silence alarm» Silence duration : between
2 alarm bips (0 to 5 seconds)[Par 2] So
[Par 3] In
[Par 4] M
[Par 5] Sy
[Par 6] Sy
[Par 7] In
[Par 8] Em
[Par 9] Bo
[Par 10] W
[Par 11] B
[Par 12] U
[Par 13] Mdisconne[Par 14] B
[Par 15] Sdisplay [Par 18] N! manual mon
auto-mre-activatecycleMaint. Maintena_INT 19
und level 7 available audible levels.itial rate If option
selected: on turning on the pump, the initial rate will bethe last selected during the last infusion. If option not
selected : 0.00 ml/h display.aximum rate Per syringe capacity (50cc / 30cc )ringe selection
Auto confirmation of the syringe or not(available only with a single syringe selected — refer to [Par
6])ringes Available syringe list (activation/disactivation).fusion start Mandatory prime or advised primepty syringe «OK»
flashes at end of infusion pre-alarm or alarm. If confirmedthe infusion goes on until the syringe is emptylus rates Per
syringe capacity (50cc / 30cc ).ard name Ward name (press OK for
each letter or — until reaching the lastposition).io name Biomedical name (press OK for each letter or —
until reachingthe last position).ser code User code: mandatory code to modify
user optionsains supplyction alarm Warning beep and message «device operating on
battery» when the pump is turned on.attery life Maximum battery life mode: allow the increase of the
battery autonomy.yringe/ward Syringe brand and capacity or ward name display.
ight modeode has priority
ode. Auto-mode d next night
Screen brightness low
Green lights low Key beep off
Manual mode: manual switch from one mode to another Auto mode:
automatic switch from one mode to another according to the time
range settingsnce Code: please contact our technical team.
tamnguyenHighlight
-
20
8. Drugs
Introduction The «drug» function makes the infusion safe,
incorporating the drug parameters. The drug library can be created
with our Vigilant Drug’Lib system which makes drug administration
safe.Vigilant Drug’Lib in particular allows the drug concentration,
default flow rate, «hard» flow rate limits (limits whichcannot be
exceeded during the infusion) and «soft» flow rates limits (limits
which can be exceeded during theinfusion after a user warning
message) the permitted infusion modes (Ml/h, V/T) and the bolus
parameter settingto be added.Start-up: Start-u
3 — No drug sele
No drug : Select «no drug» Ok: Press . set up procedure (page
10)Select drug Select the drug. Ok: Press to confirm the choice of
drug.nal) 5 — Select flow rate/start
ion to the syringe.ent is not connected.ress the key twice:
one continuous press until all ted from the line.
release the key.t.the occlusion pressure level is lue (900
mmHg).Select the flow rate with the increment keys. Check the infusion
parameters (syringe drug name and parameters, flow rate etc.) Start
Press to start the infusion. -
2309-11_nu_injectomat_agilia
Infusion mode: Volume/time and limit volumeThe infusion with a
drug name and associated parameters can also be programmed in
Mode/Time or LimitVolume mode.Start-up: Start-up procedures 1 and 2 page 10.
3 — Drug selecti
Select drug : Select the drug. Ok: Press to access and
rameters.TION> to confirm the
Check the infusion parameters (syringe, drug name and
parameters, flow rate etc.) Start: Press to start the infusion. -
22
Access to information in the drug library
Drug library Press
-
2309-11_nu_injectomat_agilia
Display and symbols
Safety feature
Service Option
Menu Access to the drug library Access to the drug library.
Drug information «EYE»: Access to the drug information
Access to drug name, concentration and drug comments which are
modifiable withSoft limit
Reset of Volume Infused and V/T and VL Parameters
OptionService(code 0200)
Vigilant
Drug lib
_INT 23
s and warnings
s
Vigilant Drug’Lib.
Flow rate limit that is allowed to be exceeded after a warning
and being validated by the user.Upper soft limit exceeded Warning message that upper soft limit
has been exceeded.Lower soft limit exceeded Warning message that lower soft limit
has been exceeded.Confirmation that the soft limits have been passed WARNING
The user must confirm that he/she is authorizing passing the
soft limit.Warning message WARNING
Drug changed CLEAR: VI and VL
If a drug or concentration of drug is changed, the volume
infused and V/T and VL parameters are reset.Function Choice Druglib [Par16] Selection Activation of drug
functions andparameters. Storage Memorize last drug used when
device was switched on.rary [Par17] Library stored in the device
and modifiablewith Vigilant Drug ‘Lib.
High flow rate
Low flow rate
-
24
Drug libraryOption Function Type of option
Loaded library
Loading of a library is acknowledged when the instrument is
switched on or a syringe is changedVigilant Drug’Lib
Library pwhich arwith Vigi
Error messages
The devi»Error mdrug
librcompatibdevice2309-11_nu_injectomat_agilia_INT*
Ok: Press to confirm change of new drug library.(*) WARNING: The
library must be changed after disconnecting the device from the
patient.arameters e modifiable lant Drug ‘Lib
The drug library must be created using Vigilant Drug’Lib, the IV
Medication Safety Solution by Fresenius Kabi. This software allows
you to adjust the dilution, the authorised and default infusion
modes, the flow rate and bolus limits (in ml/h) and can manage up
to 50 drugs.ce will display essage» if the ary is not le with the
These messages should not appear during normal operation. They
allow the instrument to self-check consistency between the Vigilant
Drug’ Lib parameters and the instrument infusion parameters.For
example: If the instrument incorporates a flow rate limit of 1500
ml/h, which is not compatible with its maximum performance (1200
ml/h), the instrument will display an error message.tamnguyenHighlight
-
2309-11_nu_injectomat_agilia
9. User test
This protocol allows a quick check of pump functionality.
Injectomat Agilia serial number
(ID/N):_____________________________Name: ________________________________Ward:
________________________________Date:
________________________________Check the state of upside down), presen Connect the device key:-
check the good funcluminous indicators.- functioning on mains Open
the syringe bsyringe).Install a 50cc syrinpusher in infusion posConfirm the syringe
an- the infusion in progre Open the syringe balarm activated.Close the syringe bDisengage and move and plunger head
alarReturn pusher to infus Note the stopper p5 ml bolus: Check the
0.5 ml. Disconnect mains OFF.The battery symbol inSignature_INT 25
Actions YES NO the device: absence of impact marks and noises
(turn the device ce of all labels as well as their legibility,
mains lead. to the mains and press thetionality of the display and
signaled by:
arrel clasp (do not install the
ge — syringe barrel clasp and ition.d select a flow rate of 0.1
ml/h.ss is signaled by man or moon.or
arrel clasp: syringe installation
arrel clasp.pusher backward. Disengagement ms activated (visible
on schemes). ion.osition/syringe volume and start a syringe stopper has moved to
5 ml BOLUS lead, the mains indicator turnsdicates a functioning on battery.
The device is operational when all the controls are OK.
Test OK
-
26
10.Performances
Rates range
Volume Limit
Volume/TimeFlow rate calculation programmed infusion calculated
at 0.00001Syringes (ml) Infusion rateInfusion rate increment: 0.1
ml/h.Indicated value corresponds to the initial device
configuration and can be changed.Bolus rate50/60 30 20 10 5
Infusion rate (ml/h) 0.1 to 2000.1 to 120
0.1 to 120
0.1 to 60
0.1 to 60
Bolus rate (ml/h)
Prime rate (ml/h)
Volume Limit (ml)
Volume to infuse
Infusion duration2309-11_nu_injectomat_agilia_INT
at volume/time programming: displayed flow rate = programmed
volume to infuse /duration, the flow rate is displayed rounded off
at 0.05 ml/h. The real flow rate is ml/h.Bolus rate increment: 50 ml/h.50 to 1200
50 to 600
50 to600
50 to350
50 to250
1200 600 600 350 250
Syringes (ml) 0.1 ml increments.KVO (keep vein open) rate: from
0.1 ml/h to 5 ml/h, stop or selected flow rate (continuous)
depending on the device configuration. Note: if KVO rate exceeds
the selected flow rate the device infuses at the selected flow
rate.50/60 30 20 10 5
From 0.1 to 999.9
Syringes (ml) 0.1 ml increments.KVO (keep vein open) rate: from
0.1 ml/h to 5 ml/h, stop or selected flow rate (continuous)
depending on the device configuration. Note: if KVO rate exceeds
the selected flow rate the device infuses at the selected flow
rate.50/60 30 20 10 5
From 0.1 to 99.9 (with 0.1 ml increments)
From 0h01 to 96h00 (with 0h01 increments)
tamnguyenHighlight
tamnguyenHighlight
tamnguyenHighlight
-
2309-11_nu_injectomat_agilia
Syringe listInjectomat Agilia offers maximum 50 syringes of
different types, brands and sizes.Accuracy
Programmable
Brand and type Syringe capacity (ml) This syringe list is
indicative of most current product codes. To know the exact list of
your product code, please contact our Sales50/60 30 20 10 5BD PLASTIPAK BD PLASTIPAK WWD BD PERFUSION
BRAUN OMNIFIX
BRAUN PERFUSOR
FRESENIUS INJECT
FRESENIUS MED. CA
FRESENIUS P-SPRIT
MONOJECT
TERUMO
Flow rate accuracy (
Device accuracy
Syringe accuracyAccuracy with back pressure of 13.33 k
Programmable paus_INT 27
pause
Department. This information can be checked directly in Ward
Option [Par 6], page 19.CAUTION: Fresenius Kabi cannot accept any responsibility for
errors in flow due to modifications of the specifications of the
syringes introduced by the manufacturer.OMAT RE ZE
*) 3 %(*) with selectable syringes, following NF EN/IEC
60601-2-24 standard.1 % 2 %
Pa 3 %
e From 1 minute to 24 h 1 minute increments.
tamnguyenHighlight
tamnguyenHighlight
tamnguyenHighlight
-
28
Pressure management (refer to user option [Util 4])
Variable modeMaximum pressure From 500 to 900 mmHg
50 mmHg increments.Defines the authorized maximum pressure
during infusion.Limit From 100 to maximum — — — : memorization of the
pressure3 levels mode
DPS(Dynamic Pressure System)2309-11_nu_injectomat_agilia_INT
limit at the device switch OFF.High From 250 to 900 mmHg
— — — : memorization of the limit (high/middle/low) at the
device switch OFF.Middle From 150 to 700 mmHg
Low From 50 to 300 mmHg
Limit Low, middle, high determined level valuesPressure increase
Anticipates an occlusion during infusion.Pressure decrease
A pressure decrease indication may be a warning of
disconnection.Drop threshold From 100 to 500 mmHg Threshold — — -:
Deactivation ofpressure decrease management.Increase threshold From 100 to 200
mmHgAccuracy: the accuracy on the pressure threshold activation is
75 mmHg or 15%.Note: 1 bar = 750 mmHg = 1000 hPa.tamnguyenHighlight
-
2309-11_nu_injectomat_agilia
Occlusion alarm response time and Bolus volume at occlusion
releaseDevice accuracy is linked to the syringe used. Values are
representative of used syringes during trials and aregiven as
indicators.Syringes used: B-D Plastipak Luer Lok.B-D Plastipak and
Luer Lok are registered trademarks of Becton Dickinson.Extension
sets used: tyNote: No other pressu= Mean = Standard deviation
Syringe
50 ml
1
5
2
20 ml
1
5
2
Syringe
50 ml5
2
20 ml5
2
m_INT 29
pe SE1400Sre measuring device was connected.
RateOcclusion alarm threshold
Values are calculated from 10 to 20 measures.
100 mmHg 500 mmHg 900 mmHg
ml/h = 25 = 4 = 1h10 = 5
= 1h40 = 10
ml/h = 415 = 50 = 12 = 2
= 20 = 4
0 ml/h = 45 = 15 = 240 = 20
= 430 = 40
ml/h = 12 = 2 = 25 = 4
= 40 = 6
ml/h = 130 = 20 = 440 = 50
= 7 = 1
0 ml/h = 20 = 5 = 50
= 11 = 130 = 20
RateBolus volume at occlusion release
Values are calculated from 20 measures after completion of the
automatic anti-bolus function.100 mmHg 500 mmHg 900 mmHg
ml/h = 0.04 ml = 0.02 ml = 0.1 ml
= 0.04 ml = 0.15 ml
= 0.05 ml0 ml/h = 0.04 ml = 0.016 ml
= 0.11 ml = 0.04 ml
= 0.15 ml = 0.07 ml
ml/h = 0.06 ml = 0.017 ml = 0.14 ml
= 0.07 ml = 0.25 ml
= 0.08 ml0 ml/h = 0.05 ml = 0.015 ml
= 0.12 ml = 0.06 ml
= 0.16 ml = 0.07 ml
m m m
m m m
m m m
m m m
m m m
m m m
m m m
m m m
m m m
m m m
-
30
11.Technical characteristics
Electrical powers Use the main lead supplied with Injectomat
Agilia.Battery Disconnect battery
Parameters are storedthis can be update by u
CommunicatioThe connector situatedpower and nurse call c
Infrared commInjectomat Agilia is einformation with the AgThe
information can thCompliance
Mains power Mains supply : 100 V — 240 V ~ / 50-60 Hz with
functional earth.Maximum consumption: 180 mAMaximum power 15 VA
External power
CharacteristicsWeightBattery life
Battery recharge
Nurse callSerial cableExternal powerPower output
Safety of ElectroMedEquipmentsEMC
(ElectroMagnetCompatibility)2309-11_nu_injectomat_agilia_INTbefore opening device. Avoid short circuits and excessive
temperatures. in the device flash memory. If the battery is totally
discharged, the date may be lost butser following mains power
connection.n port at the back of the device allows different functions
using the communication, mainsables.
unicationquipped with an infrared cell located at the back of
the device. It permits exchange ofilia Link+ rack.en be transmitted
by dedicated communication cables.consumption:Protective fuses: T2AH 250 V included in power
supply.9 Volts continuous / Power > 15 Watts.Via a specific
Fresenius Kabi accessory connected to an 8 pins connector.6 V 1.8 Ah — NiMH battery.Approximately 140 gMinimum 10 h at a
rate of 5 ml/h.Minimum 5 h at a rate of 120 ml/h.Pump OFF: < 5
h. Pump ON: < 15 h.Nurse call relay output command.TTL output.9 V / 15 W input.5 V
/ 150 mA to power Nurse Call or Serial Link accessories.Conform to the 93/42/CE Medical Directive.
IP22 Protection against splashing liquid.
Protection against leakage current:Defibrillation-proof type CF
applied part.Protection against electric shocks: class II.
Functional earth.
ical Conform to EN/IEC 60601-1 and EN/IEC 60601-2-24.
ic Conform to EN/IEC 60601-1-2 and EN/IEC 60601-2-24.
tamnguyenHighlight
tamnguyenHighlight
-
2309-11_nu_injectomat_agilia
Dimensions — Weight
Trumpet curveTrumpet curves demoncombination.The test protocol
usedplease refer to this pubThis graph is therefore overall
performance.H / L / W 135 x 345 x 160 mm
Weight around 2.1 Kg
Screen size 70 x 35 mm
Tru
O
% v
ariance fro
m
no
min
al flo
w r
ate
O
% v
ariance fro
m
no
min
al flo
w r
ate
O
% v
ariance fro
m
no
min
al flo
w r
ate_INT 31
sstrate the evolution of the minimum and maximum variance of the
Syringe/Syringe-Pumpto obtain these results is described in the EN/IEC 60601-2-24.
For further information,lication.representative of syringes used
during trials and serve as an indication only of the pump’smpet curves Start-up and instantaneous nominate curves
Used syringes: B-D Plastipak 50 ml Luer Lok.
bservation window (minutes)
Flow rate 1 ml/h
Maximum authorized %
Minimum authorized %
Max
Min
bservation window (minutes)
Flow rate 5 ml/h
Maximum authorized %
Minimum authorized %
Max
Min
bservation window (minutes)
Flow rate 20 ml/h
Maximum authorized %
Minimum authorized %
Max
Min
Flo
w r
ate
(m
l/h
) Flow rate 1 ml/h
Duration (minutes)
Flo
w r
ate
(m
l/h
) Flow rate 5 ml/h
Duration (minutes)
Flo
w r
ate
(m
l/h
) Flow rate 20 ml/h
Duration (minutes)
-
32
12.Guidance and manufacturers declaration on EMC
Electromagnetic emissions — Table 201Injectomat Agilia is
intended for use in the electromagnetic environment specified
below. The user ofInjectomat Agilia should make sure it is used in
such an environment.ElectromagneInjectomat Agilia is inThe user of Injectomat
Emissions testCompliance
obtained by the device
Electromagnetic environment — guidance
RF emissionsCISPR 11
Group 1 Injectomat Agilia uses RF energy only for its internal
function. Therefore, its RF emissions areRF emissionsCISPR 11
Harmonic emissionsIEC 61000-3-2
Voltage fluctuationsFlicker emissionsIEC 61000-3-3
D
Immunity testIE
IE
Electrostatic Discharge (ESD)IEC 61000-4-2
Electrical fastTransient / burstIEC 61000-4-4
2
for in
SurgeIEC 61000-4-5 dif
co
Voltage dips, short interruptions and voltage variations on
power supply input linesIEC 61000-4-11(>
(6
(3f
(>
Power frequency (50/60 Hz)magnetic fieldIEC 61000-4-8
Note: Ut is the a/c. main
volta2309-11_nu_injectomat_agilia_INTtic immunity — Table 202tended for use in the electromagnetic
environment specified below. Agilia should make sure it is used in
such environment.very low and are not likely to cause any interference in nearby
electronic equipment.Class B Injectomat Agilia is suitable for use in all
establishments, including domestic and hospitalestablishments and those directly connected to the public
low-voltage power supply network that supplies buildings used for
domestic purposes.Class A
oes not apply
C 60601-1-2C 60601-2-24Test level
Compliance level obtained by the
deviceElectromagnetic environment — guidance
8 kV contact 15 kV air
8 kV contact 15 kV air
Coatings of the floors out of wooden, tilling, and concrete,
with a relative humidity level at least 30 %, make it possible to
guarantee the level of necessary conformity. If it is not possible
to guarantee this environment, additional precautions must be
taken, such as: anti-static material usage, preliminary user
discharge and the wearing of anti-static clothing.kV for power supply lines
1 kV put output lines
2 kV for power supply lines
1 kV for input output lines
Mains power quality should be that of a typical domestic,
commercial or hospital environment.1 kV ferential mode
2 kVmmon mode
1 kV differential mode
2 kV common mode
Mains power quality should be that of a typical domestic,
commercial or hospital environment.< 5 % Ut95 % dip in Ut)for 0.5 cycle
< 5 % Ut(> 95 % dip in Ut)
for 0.5 cycle
Mains power quality should be that of a typical domestic,
commercial or hospital environment.For short and long interruptions (< than battery life) of
power mains, the internal battery provides the continuity of
service.40 % Ut0 % dip in Ut)for 5 cycles
40 % Ut(60 % dip in Ut)
for 5 cycles
70 % Ut0 % dip in Ut)or 25 cycles
70 % Ut(30 % dip in Ut)
for 25 cycles
< 5 % Ut95 % dip in Ut)
for 5 sec
< 5 % Ut(> 95 % dip in Ut)
for 5 sec
400 A / m 400 A / m If necessary, the power magnetic field
should be measured in the intended installation location to assure
that it is lower than compliance level.If the measured field in the
location where the Injectomat Agilia is used exceeds the applicable
magnetic field compliance level above, the Injectomat Agilia should
be observed to verify normal operation. If abnormal performance is
observed, additional measures may be necessary, such as
re-orienting or re-locating Injectomat Agilia, or install magnetic
shielding.ge prior to application of the test level.
-
2309-11_nu_injectomat_agilia
Electromagnetic immunity — Table 204Injectomat Agilia is
intended for use in the electromagnetic environment specified
below. The user of InjectomatAgilia should make sure it is used in such an environment.
Note 1: At 80 MHz and 8Note 2: these guidelines
electromagnetic
(a) Field strengtmobile radiosaccuracy. Tosurvey shoulexceeds
the operation. If re-locating In(b) Over the freq
Immunity testIEC 60601-1-2
IEC 60601-2-24Test level
Compliance level obtained by the
deviceElectromagnetic environment — guidance
Conducted RFIEC 61000-4-6
10 Vrms150 k
10 Vrms Portable and mobile RF communications equipment should
be used noRadiated RFIEC 61000-4-3
10 V80 M_INT 33
00 MHz, the highest frequency range applies.may not apply to all
situations. Absorption and reflection from structures, objects and
people affect propagation.hs from fixed transmitters, such as base stations for radio
(cellular/cordless) telephones and land, amateur radio, AM and FM
radio broadcast and TV broadcast cannot be predicted theoretically
with access the electromagnetic environment due to the fixed RF
transmitters, an electromagnetic sited be considered. If the
measured field strength in the location where Injectomat Agilia is
usedapplicable RF compliance level above, Injectomat Agilia should
be observed to verify normalabnormal performance is observed, additional measures may be
necessary, such as re-orienting orjectomat Agilia, or install
magnetic shielding.uency range 150 kHz to 80 MHz, field strengths
should be less than 10 V/m.Hz to 80 MHz closer to any part of the Injectomat Agilia
including cables, than the recommended separation distance
calculated from the equation applicable to the frequency of
transmitter.Recommended separation distance:D = 0.35 P, for a frequency of
150 kHz to 80 MHzD = 0.35 P, for a frequency of 80 MHz to 800 MHzD
= 0.7 P, for a frequency of 800 MHz to 2.5 GHzWhere P is the maximum output power rating of the transmitter in
Watts (W) according to the transmitter manufacturer and D is the
recommended separation distance in meter (m)./mHz to 2.5 GHz
10 V/m
Field strengths from fixed RF transmitters, as determined by an
electromagnetic site survey (a), should be less than compliance
level. (b)Interference may occur in the vicinity of equipment
marked with the following symbol: -
34
Recommended separation distances between portable and mobileRF
communication equipment and Injectomat Agilia — Table 206Injectomat
Agilia is intended for use in an electromagnetic environment in
which radiated RF disturbances arecontrolled. The user of
Injectomat Agilia can help prevent electromagnetic interference by
maintaining aminimum distance between portable and mobile RF
communications equipment (transmitters) and InjectomatAgilia as
recommended below, according to the maximum output power of the
communication equipment.For transmitters rated at(m) can be estimated uspower rating of
the transNote 1: At 80 MHz and 8Note 2: These guidelines
electromagnetic
The use of accesimmunity of the dThe device shoube monitored to
vcable).Rated maximumpower of trans
(W)
0.01
0.1
1
10
1002309-11_nu_injectomat_agilia_INT
a maximum output power not listed above, the recommended
separation distance d in metersing the equation applicable to the
frequency of transmitter, where P is the maximum outputmitter in
watts (W) according to the transmitter manufacturer.00 MHz, the separation distance for the highest frequency range
applies. may not apply to all situations. Absorption and reflection
from structures, objects and people affect propagation.sories and cables, other than those specified, can result in
increased emissions or decreased evice.ld not be used adjacent to other equipment. However, if adjacent
use is necessary, the device should erify normal operation in the
configuration in which it will be used (pump with a mains cable, an
RS232output mitter
Separation distance according to frequency of transmitter in
meters (m)150 kHz to 80 MHzd = 0.35 P
80 MHz to 800 MHzd = 0.35 P
800 MHz to 2.5 GHzd = 0.7 P
0.04 0.04 0.07
0.11 0.11 0.22
0.3 0.3 0.7
1.1 1.1 2.2
3.5 3.5 7
-
2309-11_nu_injectomat_agilia
13.Cleaning and use conditions
Cleaning and disinfecting Injectomat Agilia is part of the
patients immediate environment. It is advisable to clean and
disinfect thedevices external surfaces regularly and especially
before connecting a new patient and before any maintenanceoperation
in order to protect patient and staff.1. Prepare the detergent-disinfectant solution.2. Disconnect the
device from the power supply.3. Moisten the disposeach stage of the c4. Start by cleaning th
the mobile parts. P5. Continue the clean6. Clean the keyboar7.
Complete the clean8. Do not rinse, leave9. Protect and keep t10. Validate the mainte Do not place in an A Do not use:
TRICCHLORINE and AROCETONE. These aggre Take care also withcracks
in the plastic, anbe used, in accordancethe accumulation of the
Please contact theestablishment for furthEnvironmentaThe device should bedisconnected via the
btechnician.Use of the inteThis device is providedswitches to battery
moBefore starting for the without using the devicThe maximum life
of thIn case of frequent mathe device on the batteStorage conditionTemperature: — 10C tPressure : 500 hPHumidity :
10% to _INT 35able cloth with the detergent-disinfectant solution, carefully
wring out the cloth. Repeat at leaning process.e bottom side of the
device. Then carefully turn the device upside down without touching
ut down the device on a clean surface.ing on sides of the device
without wetting the sockets.d.ing of the most exposed surfaces, the
most critical zones and the mains cord.to dry.he device clean before reuse.nance protocol by simple
bacteriological checking.UTOCLAVE nor IMMERSE the device. Do not
let liquids enter the devices casing.HLOROETHYLENE-DICHLOROETHYLENE — AMMONIA — AMMONIUM CHLORIDE
-MATIC HYDROCARBON — ETHYLENE DICHLORIDE-METHYLENE CHLORIDE -ssive
agents could damage the plastic parts and cause device
malfunction.ALCOHOL BASED SPRAYS (20% — 40% alcohol). They lead to
tarnishing of and smalld do not provide the necessary cleaning
prior to disinfecting. Disinfecting SPRAYS may with the
manufacturer recommendation, from a distance of 30 cm of the
device, avoid product in liquid form. appropriate service,
responsible for cleaning and disinfecting products, in yourer details.
l conditions stored in a dry and cool place. In case of
prolonged storage, the battery should beattery access flap situated
underneath the device. This should be done by a qualifiedrnal battery with a NiMH battery. When the device is
disconnected from the main, it automaticallyde. first time, charge the battery for approx. 5 hours by
connecting the power supply corde. e battery achieved after several
charge/discharge cycles.in operations, battery life may be
decrease. To limit this risk, it is recommended to usery mode,
approximately every 4 weeks, until getting a PRE-ALARM BATTERY
signal.s and carryingo +60C.a to 1060 hPa.90%, no condensation
Use conditionsTemperature: 5C to 40C.Pressure : 700 hPa to 1060
hPa.Humidity : 20% to 90%, no condensation.tamnguyenHighlight
-
36
Recommendations Fresenius Kabi will not be liable for any
damages or claims, medical or otherwise, of any nature
whatsoever,whether direct or consequential, caused by improper use
of this device. Use only 3 parts syringes from the preprogrammed
syringes type list on the device, otherwise the specifiedaccuracy
and functioning level can not be guaranteed. Use only sterile
catheter extensions, which can resistpressures of up to 2000 hPa.
Use of certified syringes according to international standards
avoid introduction ofair in the syringe. Use of a syringe not
corresponding to one selectable on the device means that accuracy
levelscannot be guaranteed. The use of non-screflow rates and/or
high pand good medical pracprecaution should be (e.g. syringes,
extensio While in use, negatthe injection site or by High
depression maused (possible leakage Pressure variation minfusion
system charabacklash. Anti-siphon vsyringe with a line not Do not
use in conjuthan 2 000 hPa suscep Fresenius Kabi repressure
infusion devic If there is no one winfusion, this will makeand
could result in accwhich could later be infreleased. Place the
connectioas near to the start of tspace and consequentline. When
the device iattention to correctly sethe extension set is cla In
order to ensure turned ON when connfor a while, use the
Pa2309-11_nu_injectomat_agilia_INTwable extension lines or syringes may result in spillage if
infusions are carried out at highressure. Connect the infusion line
in accordance with procedures in your establishmenttices. Fresenius
Kabi recommends the use of Luer Lok type infusion lines.
Standardtaken to prevent contamination or injuries while discarding the
associated disposablen sets, needles, etc.).ive pressure variation may occur in the syringe, by the relative
height from the device tocombined infusion devices such as blood
pump, alternative clamp, etc.y create syringe siphoning. In this
situation, you must check the integrity if the syringe), and if
necessary insert anti-siphon valves.ay generate flow discontinuity
mainly noticeable at low flow rates and depending of thecteristics such as friction force, stickiness, compliance of
syringes and mechanicalalves will also eliminate any risk of free
flow during syringe changes. An air leakage in aequipped with an anti-siphon valve may generate an uncontrolled
flow delivery.nction with positive pressure infusion devices that
could generate back pressure highertible to damage infusion
disposable and the device.commends the use of one way valves or
positivees for multi-line infusions.ay valve on a gravity infusion
line during a multi-line it impossible to detect occlusions on the
patient side,umulation of the drug being infused in the gravity
line,used in an uncontrolled manner when the occlusion isn between the feeder line and the syringe-driver linehe catheter
as possible in order to minimize the deadly the impact of any
change in flow rate on the feeders placed higher than the injection site, please paycure the
syringe and manipulate the syringe only whenmped or disconnected
from patient side.all the safety features, the pump must always
remainected to the patient. Should the pump not being useduse
function.Anti-reflux valve
DO NOT USE THIS ASSEMBLY
PREFERRED INSTALLATION
-
2309-11_nu_injectomat_agilia
14.Services
Conditions of guaranteeFresenius Kabi guarantees that this
product is free from defects in material and workmanship during
theperiod defined by the accepted sales conditions, except for the
batteries and the accessories.To benefit from the materials and
workmanship guarantee from our After-Sales Service or agent
authorized byFresenius Kabi, the following conditions must be
respected: The device must have been used according to the
instructions in this Operators Guide. The device must nohandling.
The device must no The serial number ( In case of non-respparts and
labor require When return and reDepartment.Quality controUpon the hospital requA regular control
checTechnical manual. Theby any contract or agrePreventive maTo ensure normal perevery 3 years. This incThe
qualified techniciadropped or if any of maCAUTION: Failure to
functional failure. Internto the pump or user.ServicingFor further informatioCustomer service.If a device is
returnedcarefully, if possible inFresenius Kabi is noAt the end of the delectrical and electroncompetent recycling
o_INT 37t have been damaged when in storage, at the time of repair, or
show signs of impropert have been altered or repaired by non-qualified personnel.ID/N)
must not have been altered, changed, or erased.ect of these
conditions, Fresenius Kabi will prepare an estimate for repair
covering thed.pair of a device is necessary, please contact
Fresenius Kabi Customer or After-Saleslest, a control check of the device may be performed every 12
months.k (not included in the guarantee) consists of various
inspection operations listed in these control checks must be
performed by an experienced technician and are not coveredement
provided by Fresenius Kabi.intenanceformance of the device, it is recommended that
preventive maintenance is performedludes battery replacement and it
should be performed by a qualified technician.ns in your
establishment or our After-Sales Service should be informed if the
device islfunctions occurs. In this case, the device must not be
used.comply with these maintenance procedures can damage the device
and lead to aal inspection of the device requires the respect of
particular procedures to void damagesn concerning the device servicing or use, please contact our
After-Sales Service or ourto our After-Sales Department, it is essential to clean and
disinfect it, then, pack it very its original packaging, before
sending it.t liable for loss or damage to the device during transport to
our After-Sales Department.evice life, return it to an organization competent in the
treatment of the ic equipment waste. Remove the battery from the
device and return it to a rganization. -
38
Data racks, accessories and maintenance toolsInjectomat Agilia
is compatible with the Agilia accessories range.For further
information, please contact our Commercial Department.Data managemen
Maintenance CD
Vigilant, the IV M
Ref.
Duo Agilia 2 channels accessory for power supply centralisation
073495Y Duo Agilia cable
DC-DC converter Agili
Nurse call Agilia
Link 4 Agilia
Link 6 Agilia
Link 8 Agilia
Link 4 + Agilia
Link 6 + Agilia
Link 8 + Agilia
RS 232 cable for Agilia
USB cable for Agilia
Vigilant Ethernet cable
Vigilant WIFI cable for
Partner Agilia
Maintenance kit Agilia
Vigilant Drug Lib for 2309-11_nu_injectomat_agilia_INT
t
& tools
edication Safety Solution
2 channels cable for DC/DC power centralisation 073497
a Cable for transportation (ambulances) 073494
Nurse call cable (4000 V isolated) 073496
Rack 4 slots for power centralisation 073480
Rack 6 slots for power centralisation 073481
Rack 8 slots for power centralisation 073498
Rack 4 slots for power centralisation and communication
capabilities073482
Rack 6 slots for power centralisation and communication
capabilities073483
Rack 8 slots for power centralisation and communication
capabilities073499
Communication cable for RS232 connection (4000V isolated)
073493Communication cable for USB connection (4000V isolated)
073491for Agilia Communication cable for Ethernet connection(4000V
isolated)073490
Agilia Communication cable for WIFI connection (4000V isolated)
073492Maintenance CD 067037
Maintenance tool box 178950
Agilia Software for drugs adjustment 073473
-
2309-11_nu_injectomat_agilia
This user guide may contain inModifications may thus be
maBecause of the evolution of theapplicable only for the device
This user guide may not be reregistered trademarks in the nRevision date: July 2010_INT 39
accuracies or typographical errors. de and will be included in
later editions. standards, lawful texts and material, the
characteristics indicated by the text and the images of this
document arethat it accompanies.produced in whole or in part without the
written consent of Fresenius Kabi. Injectomat, Vigilant and Agilia
are ame of Fresenius Kabi in selected countries.Fresenius Vial S.A.SLe Grand CheminF-38590
Brzinswww.fresenius-kabi.com -
2309
-11_
nu_i
njec
tom
at_a
gilia
_IN
T
Injectomat AgiliaIntroductionIntended usePrecautions to be
takenDescription of Injectomat AgiliaOperations for useDisplay and
symbolsAlarm and safety featuresMenuPermanent menuMenu to be
selected in option modeOptionDrugsIntroductionInfusion mode: Volume/time and limit
volumeDisplay and symbolsSafety features and warningsService
OptionsDrug libraryUser testPerformancesRates rangeVolume LimitVolume/TimeSyringe
listAccuracyProgrammable pausePressure managementOcclusion alarm
response time and Bolus volume at occlusion releaseTechnical characteristicsElectrical powersBatteryCommunication
portInfrared communicationComplianceDimensions — WeightTrumpet
curvesGuidance and manufacturers declaration on EMCElectromagnetic
emissions — Table 201Electromagnetic immunity — Table
202Electromagnetic immunity — Table 204Recommended separation
distances between portable and mobile RF communication equipment
and Injectomat Agilia — Table 206Cleaning and use conditionsCleaning and
disinfectingEnvironmental conditionsUse of the internal
batteryRecommendationsServicesConditions of guaranteeQuality controlPreventive
maintenanceServicingData racks, accessories and maintenance
tools
-
Главная
-
Каталог
-
Реанимация и анестезиология
-
Инфузионные насосы
- Инфузионный насос Fresenius Injectomat MC Agilia
Модель шприцевого насоса Injectomat MC Agilia имеет расширенный набор функций (программирование скорости введения дозы, введение нагрузочной дозы, программирование болюса, графическая история и др.), предназначен для использования в отделениях интенсивной терапии и других специализированных отделениях.
- Артикул
- 0640709369
- Производитель
- Fresenius
- Страна производства
- Франция
- Гарантия производителя
- 1 год
- Срок поставки
- Под заказ


Технические характеристики инфузионного насоса Fresenius Injectomat MC Agilia
| Состояние | Новое |
|---|---|
| Тип аппарата | Шприцевой |
| Режим инфузии | Болюсная инфузия, Открытой вены KVO, С заданной скоростью |
| Количество шприцев | 1 |
| Время непрерывной работы, ч | 12 |
| Дополнительные возможности | Встроенная память результатов, Вывод воздуха из шприца |
Подробное описание инфузионного насоса Fresenius Injectomat MC Agilia
Подробное описание инфузионного насоса Fresenius Injectomat MC Agilia
Варианты программирования инфузии:
- программирование скорости в мл/ч
- программирование в единицах дозы (например, мкг/кг/мин) – насос автоматически пересчитает скорость в мл/ч
- программирование введения определенного объема или дозы за определенное время
- программирование объема и скорости инфузии
- возможность программирования введения нагрузочной дозы/дозы индукции
Преимущества:
- В память загружены характеристики 50 моделей шприцев разных производителей, что позволяет учесть их особенности и обеспечить высокую точность при проведении инфузии
- При установке шприца в насос перемещение толкателя и фиксация поршня осуществляется вручную – нет задержек времени
- Защита поршня – защитная рамка предотвращает непреднамеренное введение болюса при случайном ударе по толкателю поршня, например при падении насоса или при транспортировке
- Большой информативный экран – достаточно беглого взгляда, чтобы быстро оценить состояние инфузии
- Отражение на экране названия вводимого раствора/препарата
- Возможность установки библиотеки лекарственных препаратов Vigilant® Drug Lib. В ней для каждого препарата программируется ряд параметров: название, мягкие пределы (наиболее высокая используемая скорость введения), жесткие пределы (максимально допустимая скорость введения), допустимые режимы инфузии, что значительно повышает безопасность при проведении инфузии
- Режим поддержания вены открытой (ПВО) – после завершения инфузии насос автоматически продолжит вводить раствор с небольшой скоростью (от 1 до 5 мл/ч) с целью поддержания проходимости венозного доступа
- Динамическая система контроля давления – реагирует на стабильное изменение уровня давления в системе (до достижения заданного предела), позволяет быстро обнаружить окклюзию или отсоединение системы и своевременно принять необходимые меры
- Графическое и цифровое отражение значения давления в системе
- Графическое отражение истории: объем/введенная доза, давление, скорость
- Поворачиваемый зажим позволяет крепить насос в необходимом положении
- Возможность объединения насосов друг с другом
- Ночной режим – сниженная яркость, выключение звука кнопок; автоматический переход в ночной режим в заданное время
В этом сегменте ПМК также рекомендует
Fresenius Injectomat MC Agilia
Цена по запросу
INJECTOMAT MC AGILIA
ИНФУЗИЯ
СКОРОСТЬ ИНФУЗИИ 0.1 — 1200 мл/час в обычном режиме, в зависимости от объема шприца (0.1мл/час шаг).
0,1-9,99 мл/час в микро режиме.
ТОЧНОСТЬ ИНФУЗИИ ± 1% механизм; ± 2 % шприц.
ОБЪЕМ ШПРИЦЕВ 5, 10, 20, 30/35, 50/60 мл.
ТИПЫ ШПРИЦЕВ (ПРОИЗВОДИТЕЛЬ) До 50 типов.
РЕЖИМЫ ИНФУЗИИ • мл/час.
• Доза
Единицы дозировки: ng, μg, mg, cal, kcal, U, kU, mmol, mol, Kmol / kg.
Единицы времени: 24ч / ч / мин.
Разведение: X единиц / мл или X единиц / Y мл.
• Объем или доза / время: 0.1 — 99.9 единиц ; 00 ч 01 — 96 ч 00.
• Предел объема 0.1 — 999.9 мл.
ВВЕДЕННЫЙ ОБЪЕМ/ДОЗА Объем: 0.1 — 999.9 мл.
ПРОМЫВКА 3 режима: обязательный, не обязательный, рекомендованный / скорость:1200 мл/час.
БОЛЮС Ручной болюс: скорость: 50 — 1200 мл/час (50 мл/час шаг).
Программируемый болюс (доза или объем / время): 0.1 — 99.9 единиц / 0.1 — 1200 мл время автоматический
расчет.
ДОЗА ИНДУКЦИИ Доза или объем / время: 0.1 — 99.9 единиц / 00 мин 01 — 59 час 59 автоматический расчет скорости.
ОКОНЧАНИЕ ИНФУЗИИ (О/В &ПО) ПВО 1 и ПВО 2: 0.1 — 5 мл/час регулируемая и сохраняемая, продолжительная инфузия, остановка.
БЫСТРОЕ НАЧАЛО ИНФУЗИИ Как стандарт.
ПАУЗА Программируемая от 1 мин до 24 часов, шаг 1 минута.
ИСТОРИЯ СОБЫТИЙ 1500 последних событий.
ГРАФИЧЕСКАЯ ИСТОРИЯ Объем / доза введенная, давление, скорость..
НОЧНОЙ РЕЖИМ В ночном режиме уменьшается яркость дисплея и зеленых индикаторов. Выключение звука кнопок. Ночной
режим может быть включен непосредственно, либо запрограммирован по времени.
ПРОГРАММИРУЕМЫЕ РЕЖИМЫ Без названия лекарства: инфузия без отображения названия лекарства на дисплее.
Маркировка лекарств: инфузия с отображением названия лекарства.
Vigilant Drug’Lib: режим введения лекарств с дополнительными параметрами.
МАРКИРОВКА ЛЕКАРСТВ Вывод на дисплей одного из 50 лекарств из предустановленного списка.
БИБЛИОТЕКА ЛЕКАРСТВ Программируемая с помощью Vigilant® библиотека лекарств для повышения безопасности инфузии.
До 120 лекарств (зависит от размера комментариев) разделенных на 4 библиотеки.
УПРАВЛЕНИЕ ДАВЛЕНИЕМ
КОНТРОЛЬ ДАВЛЕНИЯ 2 доступных режима: изменяемый (от 50 до 750 мм Hg, 25-50 мм Hg шаг) либо выбор из 3 предустановленных.
ДСД Динамическая Система Давления ДСД – предупреждение об изменениях давления.
Быстрое определение повышения давления, падения давления (отсоединение магистрали, утечка).
МОНИТОРИНГ ДАВЛЕНИЯ Графическое изображение давления в инфузионной линии и выбранного предела давления в виде
пиктограммы.
СИСТЕМА АНТИ-БОЛЮС Снижение болюса после устранения причины окклюзии до 0.2 мл макс.
ТРЕВОГИ/ПРЕТРЕВОГИ/БЕЗОПАСНОСТЬ
СТАТУС Зеленые индикаторы для нормальной работы, оранжевые – претревога и предупреждения, красные – тревога.
Сигналы видны с расстояния 5 м.
Все тревоги разъясняются специальными символами , текстовыми сообщениями и звуковым сигналом.
КОНТРОЛЬ УСТАНОВКИ ШПРИЦА Контроль установки кромки шприца, определение головки поршня, проверка анти-сифонной системы, контроль
фиксации корпуса шприца.
КОНТРОЛЬ ИНФУЗИИ Претревога окклюзии, тревога окклюзии, претревога окончания инфузии, тревога окончания инфузии,
претревога достижения предела объема, тревога достижения предела объема, блок кнопок, жесткие и мягкие
ограничения скорости.
КОНТРОЛЬ НАСОСА Тревога механизма, претревога по заряду батареи, тревога разряженной батареи, время заряда батареи в часах
и минутах для выбранной скорости, не подтвержденные установки, технические тревоги с кодами (авто-тест,
вращение мотора), проверка приводящего механизма, проверка прибора, ошибка коммуникации
СЕРВИС Предупреждение о необходимости планового сервиса.
ТЕХНИЧЕСКИЕ ХАРАКТЕРИСТИКИ
ЗАЩИТА ПОРШНЯ Защита поршня шприца от механических ударов “Push-Guard”.
ЭКРАН Монохромный (голубой) LCD дисплей, размер 66 мм x 33 мм (128 x 64 пикселей).
ПОВОРАЧИВАЕМЫЙ ЗАЖИМ Поворачиваемый зажим, позволяющий крепить насос на стойку либо рельсу /стойка: 20-40мм максимум; рельса:
25-35 x 10 мм.
СОВМЕСТИМОСТЬ Возможность комбинации 3-х насосов между собой без дополнительных приспособлений.
РАЗМЕРЫ (В/Ш/Г)/ВЕС 160 x 345 x 135 мм/2150 г.
БАТАРЕЯ Батарея: NI/MH / Минимальный заряд: 10 часов при скорости 5 мл/час.
Замена батареи: независимый компонент– не требует проведения специфических тестов.
СООТВЕТСТВИЕ СТАНДАРТАМ EN/IEC 60601-1 и EN/IEC 60601-2-24 / CE 0459 маркировано в соответствии EEC 93/42 Европейская Директива по
мед. оборудованию / ЭМС.
ВЛАГОЗАЩИТА IP 22.
ЭЛЕКТРО СОВМЕСТИМОСТЬ Защита от утечки напряжения: CF тип – защита от электрошока: класс II.
ЭЛЕКТРОПИТАНИЕ 100 — 240 Vac / 15 VA, внешний источник 9 Vdc.
В наличии документация на медицинское оборудование фирмы Fresenius (в т.ч. на русском языке):
Modification: 20 October 2023
Техническая документация (Technical Documentation/Manual) на 2008K [Fresenius] Гемодиализ
Инструкция по наладке (Adjustment Instruction) на 2008K Calibration procedures [Fresenius] Гемодиализ
Техническая документация (Technical Documentation/Manual) на 2008K2 [Fresenius] Гемодиализ
Схема электрическая (Electric scheme (circuit)) на 4008 B/E/S/H [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 4008 BVM [Fresenius] Гемодиализ
Техническая документация (Technical Documentation/Manual) на 4008 E/B/H/S [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 4008B v3.2 [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 4008B v3.2 1996 [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 4008H/S v4.0 OCM Дополнительные инструкции [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 4008S [Fresenius] Гемодиализ
Сервисная инструкция (Service manual) на 5008 [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 5008 [Fresenius] Гемодиализ
Техническая документация (Technical Documentation/Manual) на 5008 (Диагностика гидравлики) [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на 5008S [Fresenius] Гемодиализ
Техническая документация (Technical Documentation/Manual) на Ionometer 2 [Fresenius] Анализаторы
Техническая документация (Technical Documentation/Manual) на Ionometer 3 [Fresenius] Анализаторы
Инструкция по эксплуатации (Operation (Instruction) manual) на Ionometer 3 [Fresenius] Анализаторы
Сервисная инструкция (Service manual) на multiFiltrate [Fresenius] Гемодиализ
Техническая документация (Technical Documentation/Manual) на multiFiltrate [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на multiFiltrate V1.9 [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на multiFiltrate V5.2 [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на Prometeus серии 4008Н «Прометей» [Fresenius] Гемодиализ
Техническая документация (Technical Documentation/Manual) на Инфузомат Pilot A, CE [Fresenius] Разное
Техническая документация (Technical Documentation/Manual) на Инфузомат Pilot A2, CE2 [Fresenius] Разное
Техническая документация (Technical Documentation/Manual) на Инфузомат Pilot C2, CE2 [Fresenius] Разное
Техническая документация (Technical Documentation/Manual) на Инфузомат Pilot Delta [Fresenius] Разное
Техническая документация (Technical Documentation/Manual) на Сепаратор клеток крови COM.TEC [Fresenius] Лаборатория
Техническая документация (Technical Documentation/Manual) на Система осмотическая Aquasafe 08 [Fresenius] Разное
Инструкция по эксплуатации (Operation (Instruction) manual) на Системы автопереливания C.A.T.S plus [Fresenius] Гемодиализ
Инструкция по эксплуатации (Operation (Instruction) manual) на Установка обратного осмоса Aqua WTU 125/250 [Fresenius] Разное
Инструкция по эксплуатации (Operation (Instruction) manual) на Устройство для обработки воды WTU 50, 100 [Fresenius] Стерилизаторы
Эксплуатационная и сервисная документация (Operating and Service Documentation) на Устройство приготовления концентрата GRANUMIX 107S/507S [Fresenius] Разное
Техническая документация (Technical Documentation/Manual) на Шприцевой дозатор Injectomat 2000 MC [Fresenius] Разное
Техническая документация (Technical Documentation/Manual) на Шприцевой дозатор Injectomat Agilia [Fresenius] Разное
Инструкция пользователя (User manual) на Шприцевой дозатор Injectomat Agilia [Fresenius] Разное
Архив технической документации медицинского оборудования © 2006 … 2023




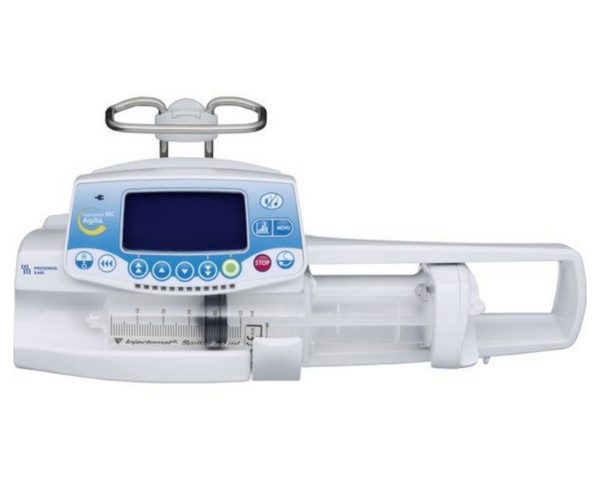



![Техническая документация Technical Documentation/Manual на 2008K [Fresenius] Техническая документация Technical Documentation/Manual на 2008K [Fresenius]](https://medtechnic.net.ua/img_d/9000/9379.jpg)
![Инструкция по наладке Adjustment Instruction на 2008K Calibration procedures [Fresenius] Инструкция по наладке Adjustment Instruction на 2008K Calibration procedures [Fresenius]](https://medtechnic.net.ua/img_d/9000/9378.jpg)
![Техническая документация Technical Documentation/Manual на 2008K2 [Fresenius] Техническая документация Technical Documentation/Manual на 2008K2 [Fresenius]](https://medtechnic.net.ua/img_d/9000/9387.jpg)
![Схема электрическая Electric scheme (circuit) на 4008 B/E/S/H [Fresenius] Схема электрическая Electric scheme (circuit) на 4008 B/E/S/H [Fresenius]](https://medtechnic.net.ua/img_d/7000/7304.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 4008 BVM [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 4008 BVM [Fresenius]](https://medtechnic.net.ua/img_d/2000/2575.jpg)
![Техническая документация Technical Documentation/Manual на 4008 E/B/H/S [Fresenius] Техническая документация Technical Documentation/Manual на 4008 E/B/H/S [Fresenius]](https://medtechnic.net.ua/img_d/9000/9015.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 4008B v3.2 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 4008B v3.2 [Fresenius]](https://medtechnic.net.ua/img_d/6000/6621.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 4008B v3.2 1996 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 4008B v3.2 1996 [Fresenius]](https://medtechnic.net.ua/img_d/6000/6429.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 4008H/S v4.0 OCM Дополнительные инструкции [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 4008H/S v4.0 OCM Дополнительные инструкции [Fresenius]](https://medtechnic.net.ua/img_d/10000/10092.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 4008S [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 4008S [Fresenius]](https://medtechnic.net.ua/img_d/5000/5705.jpg)
![Сервисная инструкция Service manual на 5008 [Fresenius] Сервисная инструкция Service manual на 5008 [Fresenius]](https://medtechnic.net.ua/img_d/7000/7361.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 5008 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 5008 [Fresenius]](https://medtechnic.net.ua/img_d/6000/6619.jpg)
![Техническая документация Technical Documentation/Manual на 5008 (Диагностика гидравлики) [Fresenius] Техническая документация Technical Documentation/Manual на 5008 (Диагностика гидравлики) [Fresenius]](https://medtechnic.net.ua/img_d/7000/7359.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на 5008S [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на 5008S [Fresenius]](https://medtechnic.net.ua/img_d/6000/6620.jpg)
![Техническая документация Technical Documentation/Manual на Ionometer 2 [Fresenius] Техническая документация Technical Documentation/Manual на Ionometer 2 [Fresenius]](https://medtechnic.net.ua/img_d/1000/1667.jpg)
![Техническая документация Technical Documentation/Manual на Ionometer 3 [Fresenius] Техническая документация Technical Documentation/Manual на Ionometer 3 [Fresenius]](https://medtechnic.net.ua/img_d/3000/3874.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на Ionometer 3 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на Ionometer 3 [Fresenius]](https://medtechnic.net.ua/img_d/5000/5624.jpg)
![Сервисная инструкция Service manual на multiFiltrate [Fresenius] Сервисная инструкция Service manual на multiFiltrate [Fresenius]](https://medtechnic.net.ua/img_d/7000/7434.jpg)
![Техническая документация Technical Documentation/Manual на multiFiltrate [Fresenius] Техническая документация Technical Documentation/Manual на multiFiltrate [Fresenius]](https://medtechnic.net.ua/img_d/6000/6138.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на multiFiltrate V1.9 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на multiFiltrate V1.9 [Fresenius]](https://medtechnic.net.ua/img_d/9000/9755.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на multiFiltrate V5.2 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на multiFiltrate V5.2 [Fresenius]](https://medtechnic.net.ua/img_d/0000/927.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на Prometeus серии 4008Н "Прометей" [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на Prometeus серии 4008Н "Прометей" [Fresenius]](https://medtechnic.net.ua/img_d/3000/3171.jpg)
![Техническая документация Technical Documentation/Manual на Инфузомат Pilot A2, CE2 [Fresenius] Техническая документация Technical Documentation/Manual на Инфузомат Pilot A2, CE2 [Fresenius]](https://medtechnic.net.ua/img_d/1000/1475.jpg)
![Техническая документация Technical Documentation/Manual на Сепаратор клеток крови COM.TEC [Fresenius] Техническая документация Technical Documentation/Manual на Сепаратор клеток крови COM.TEC [Fresenius]](https://medtechnic.net.ua/img_d/4000/4685.jpg)
![Техническая документация Technical Documentation/Manual на Система осмотическая Aquasafe 08 [Fresenius] Техническая документация Technical Documentation/Manual на Система осмотическая Aquasafe 08 [Fresenius]](https://medtechnic.net.ua/img_d/4000/4591.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на Системы автопереливания C.A.T.S plus [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на Системы автопереливания C.A.T.S plus [Fresenius]](https://medtechnic.net.ua/img_d/9000/9412.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на Установка обратного осмоса Aqua WTU 125/250 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на Установка обратного осмоса Aqua WTU 125/250 [Fresenius]](https://medtechnic.net.ua/img_d/5000/5706.jpg)
![Инструкция по эксплуатации Operation (Instruction) manual на Устройство для обработки воды WTU 50, 100 [Fresenius] Инструкция по эксплуатации Operation (Instruction) manual на Устройство для обработки воды WTU 50, 100 [Fresenius]](https://medtechnic.net.ua/img_d/2000/2574.jpg)
![Эксплуатационная и сервисная документация Operating and Service Documentation на Устройство приготовления концентрата GRANUMIX 107S/507S [Fresenius] Эксплуатационная и сервисная документация Operating and Service Documentation на Устройство приготовления концентрата GRANUMIX 107S/507S [Fresenius]](https://medtechnic.net.ua/img_d/8000/8343.jpg)
![Техническая документация Technical Documentation/Manual на Шприцевой дозатор Injectomat 2000 MC [Fresenius] Техническая документация Technical Documentation/Manual на Шприцевой дозатор Injectomat 2000 MC [Fresenius]](https://medtechnic.net.ua/img_d/4000/4225.jpg)
![Техническая документация Technical Documentation/Manual на Шприцевой дозатор Injectomat Agilia [Fresenius] Техническая документация Technical Documentation/Manual на Шприцевой дозатор Injectomat Agilia [Fresenius]](https://medtechnic.net.ua/img_d/4000/4532.jpg)
![Инструкция пользователя User manual на Шприцевой дозатор Injectomat Agilia [Fresenius] Инструкция пользователя User manual на Шприцевой дозатор Injectomat Agilia [Fresenius]](https://medtechnic.net.ua/img_d/4000/4531.jpg)